|
The electron holding capacity of each orbit is 2n 2. K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. These circular paths are called orbit(shell). The electrons of the atom revolve around the nucleus in a certain circular path. The complete idea of the orbit is given there. Scientist Niels Bohr was the first to give an idea of the atom’s orbit. Antimony atom electron configuration through orbit For example Aufbau principle, Hund’s principle, and Pauli’s exclusion principle. Electron configuration through orbital (Aufbau principle)Įlectron configuration through orbitals follows different principles.Electron configuration through orbit (Bohr principle).Electron configuration can be done in two ways. The electron configuration of antimony is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 3, if the electron arrangement is through orbitals. The arrangement of electrons in antimony in specific rules in different orbits and orbitals is called the electron configuration of antimony. These electrons are arranged according to specific rules in different orbitals. The total number of electrons in antimony is fifty-one. What is the electron configuration of antimony? Hopefully, after reading this article, you will know more about this topic. I also discussed how to draw and write an orbital diagram of antimony. In this article, I have discussed in detail how to easily write the complete electron configuration of antimony. Antimony is a classified metalloid element. 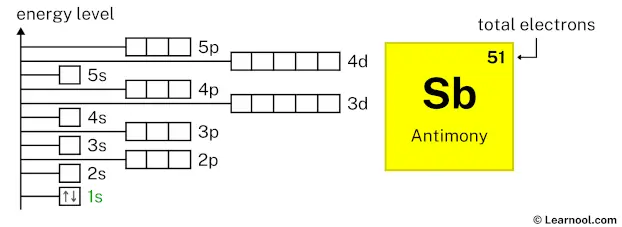
Antimony is the 51st element in the periodic table and its symbol is ‘Sb’.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
